Treatment for DCIS
Listen to our Real Pink podcast, Impact of Diagnosis on Your Mental Health.
What is DCIS?
DCIS (ductal carcinoma in situ) is a non-invasive breast cancer. It’s stage 0 breast cancer.
With DCIS, the abnormal cells are contained in the milk ducts and have not invaded nearby tissue outside the milk ducts. (The milk ducts are canals that carry milk from the lobules to the nipple openings during breastfeeding.)
It’s called “in situ” (which means “in place”) and “non-invasive” because the abnormal cells have not left the milk ducts.
DCIS is also called intraductal (within the milk ducts) carcinoma. Some people use the terms “pre-invasive” or “pre-cancerous” to describe DCIS.
Both women and men can get DCIS.
DCIS is treated to try to prevent the development of invasive breast cancer.
Learn more about breast cancer stages and staging.
Find statistics on breast cancer.
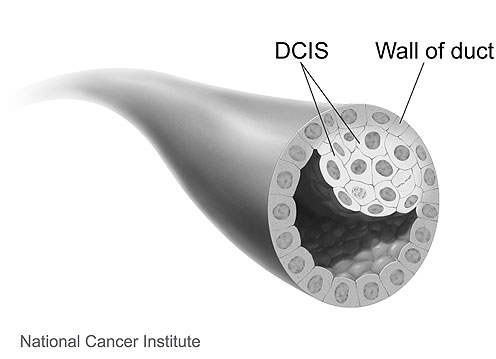 |
Image source: National Cancer Institute (www.cancer.gov) |
The following is a 3D interactive model showing DCIS. The labels show a normal duct and a duct with DCIS.
DCIS diagnosed with invasive breast cancer
DCIS can be found alone or with invasive breast cancer.
If it’s diagnosed with invasive breast cancer, treatment and the chances of survival are based on the invasive breast cancer, not the DCIS.
Learn about treatment for early breast cancer.
Treatment for DCIS
DCIS is non-invasive, but without treatment, the abnormal cells could progress to invasive breast cancer over time.
Left untreated, it’s estimated 10%-50% of DCIS cases may progress to invasive breast cancer [1-4].
Health care providers cannot predict which cases of DCIS will progress to invasive breast cancer and which will not. Because DCIS might progress to invasive breast cancer, almost all cases are treated.
Surgery is recommended as the first step to treat DCIS. After surgery, some people will have radiation therapy, and some may take hormone therapy.
Learn more about treatments for DCIS.
Learn about the risk of invasive breast cancer after treatment for DCIS.
Learn about emerging areas in the treatment of DCIS.
Watch our video of Former Komen Chief Scientific Advisor Dr. George Sledge as he shares some things to think about before getting treatment.
Your health care team
Throughout your treatment and beyond, you’ll get care from many health care providers. Your health care team may include:
- Doctors involved in cancer treatment (medical oncologists, surgeons, radiation oncologists)
- Doctors involved in other care for people with breast cancer (radiologists, pathologists, plastic surgeons and others)
- Your primary care doctor
- Nurses
- Genetic counselors
- Patient navigators
- Social workers
- Mental health providers (counselors, clinical social workers, psychologists and others)
- Dietitians
- Physical therapists
- Pharmacists
- Integrative care specialists
- Other health care providers (you’ll continue to see your gynecologist and any other specialists)
These health care providers may be involved in your care throughout diagnosis, treatment and beyond.
It’s OK to get a second opinion at any point during your care. This may help you get a different insight into your diagnosis and treatment options. Your doctor should never discourage you from getting a second opinion.
Learn more about your breast cancer care team.
Learn about choosing a doctor.
Learn about choosing a hospital.
Learn more about getting a second opinion.
Online access to your medical information
Most hospitals and doctors’ offices now allow you access to your medical information online. You set up an account with a login name and password. This provides security to protect your privacy.
An online account gives you access to your test results, pathology and radiology reports, prescription drug information, medical appointments, doctors’ notes and more. It also gives you another way to communicate with your health care team.
Your surgeon or your oncologist will go over the main findings of your pathology report (including test results) with you and answer any questions you may have. You can learn more about treatment for DCIS below.
You may see some information online before you meet with your surgeon or your oncologist. This information is written in medical language because it’s prepared for health care providers. This can make some of the wording hard to understand, but our page on the contents of a pathology report may help.
Staying organized |
|
You’ll be getting information from different members of your health care team. So, it may be helpful to use a notebook or 3-ring binder with pockets, apps on your phone or tablet, or other organizer to keep track of your breast cancer treatments and health care team. You may want to include:
Susan G. Komen® has interactive Questions to Ask Your Doctor resources and a Breast Cancer Diagnosis Discussion Guide that you can download and write on and may be helpful to include. |
Treatment guidelines
Although the exact treatment for DCIS varies from person to person, guidelines help make sure high-quality care is given. These guidelines are based on the latest research and agreement among experts.
The National Comprehensive Cancer Network (NCCN) and the American Society of Clinical Oncology (ASCO) are respected organizations that regularly review and update their guidelines.
In addition, the National Cancer Institute (NCI) has treatment overviews.
Talk with your health care team about which treatment guidelines they follow.
After you get a recommended treatment plan from your health care team, study your treatment options. Together with your health care team, make thoughtful, informed decisions that are best for you. Each treatment has benefits and risks to consider along with your own values and lifestyle.
Survival after DCIS
With treatment, the chances of survival for DCIS are usually excellent.
Learn more about breast cancer survival rates.

Thelma Brown, Komen Advocates in Science member
“A diagnosis of any stage of breast cancer can be very frightening. Often, the first instinct is to act quickly. There is not a one size fits all approach to breast cancer, and DCIS is no different. Therefore, it is important to slow down and take the time to learn about DCIS and your treatment options. This will enable you to be an active member of your health care team and share in the decision-making.”
Surgery for DCIS
Surgery is the first step to treat DCIS. It removes the abnormal tissue from the breast. It’s the main treatment for DCIS, but you may also have other treatments.
Depending on how far the DCIS has spread within the milk ducts, surgery can be a mastectomy or a lumpectomy (breast-conserving surgery).
Mastectomy for DCIS
If DCIS is spread throughout the ducts, affecting a large part of the breast, a total (simple) mastectomy will be done. With a total mastectomy, the surgeon removes the entire breast and possibly some lymph nodes, but no other tissue.
Overall survival is the same for women with DCIS who have a mastectomy and those who have a lumpectomy (with or without radiation therapy) [5].
Some women choose to have breast reconstruction to help restore the look of the breast that was removed. Reconstruction may be immediate (done at the same time as the mastectomy) or delayed (done at a later time).
If you want reconstruction, find out if there’s a plastic surgeon available who specializes in breast reconstruction. This may be important when making a decision about surgery. Discuss your reconstruction options with your plastic surgeon before your breast surgery.
Learn about deciding between a mastectomy and a lumpectomy.
Sentinel lymph node biopsy
A sentinel lymph node biopsy is sometimes done at the same time as a mastectomy for DCIS.
A sentinel lymph node biopsy is a procedure used to check whether or not invasive breast cancer has spread to the axillary lymph nodes (the lymph nodes in the underarm area). The surgeon removes 1-5 axillary lymph nodes.
In some cases, a sentinel lymph node biopsy helps some people avoid a more invasive lymph node surgery that removes more lymph nodes (called an axillary lymph node dissection).
Once a mastectomy has been done, a person can’t have a sentinel lymph node biopsy.
If it turns out there’s invasive breast cancer (along with DCIS) in the tissue removed during the mastectomy, and a sentinel lymph node biopsy wasn’t done, an axillary lymph node dissection might be needed. So, having a sentinel lymph node biopsy helps some people avoid an axillary lymph node dissection.
Lumpectomy for DCIS
If there’s little spread of DCIS within the ducts, you will likely have a choice between a mastectomy or a lumpectomy.
With a lumpectomy, the surgeon removes only the abnormal tissue in the breast and a small rim of normal tissue around it (called a margin). The rest of the breast is left intact. Lymph nodes are not usually removed with a lumpectomy for DCIS.
Overall survival is the same for women with DCIS who have a mastectomy and those who have a lumpectomy (with or without radiation therapy) [5].
In the U.S., most women with DCIS are treated with a lumpectomy followed by radiation therapy [6].
Learn about deciding between a mastectomy and a lumpectomy.
Radiation therapy
Standard radiation therapy (also called radiotherapy) uses targeted, high-energy X-rays or other forms of radiation to kill cancer cells.
The goal of radiation therapy is to kill any cancer cells that might be left in the breast after breast cancer surgery.
After a mastectomy
Women treated with a mastectomy for DCIS generally don’t benefit from radiation therapy. However, in rare cases, it may be recommended because of the extent or location of the DCIS.
After a lumpectomy
A lumpectomy for DCIS is often followed by radiation therapy to lower the risk of [5-12]:
- DCIS recurrence (a return of DCIS) in the treated breast
- Invasive breast cancer in the treated breast
A meta-analysis that combined the results of 4 randomized clinical trials showed adding whole breast radiation therapy after a lumpectomy reduced the risk of DCIS recurrence or invasive breast cancer (each in the breast treated for DCIS) by half compared to a lumpectomy alone [6].
Overall survival is the same for women with DCIS who have a lumpectomy with or without radiation therapy [5-7]. So, the recommendation for radiation therapy after a lumpectomy for DCIS has become more personalized. It’s based on factors including DCIS tumor size, DCIS features (as seen under a microscope), including tumor grade and surgical margins. It’s also based on a woman’s age and personal preferences.
Low-grade and intermediate-grade DCIS tend to grow more slowly than high-grade DCIS. Margins are negative when there are no cancer cells in the rim of breast tissue surrounding the tumor that was removed during surgery.
Some women who have a low risk of DCIS recurrence or invasive breast cancer after a lumpectomy may be recommended to have partial breast radiation therapy (radiation only to the part of the breast where the DCIS was removed) or no radiation therapy [5]:
- Women who have small, low-grade or intermediate-grade DCIS and negative surgical margins may be recommended to have partial breast radiation therapy instead of whole breast radiation therapy.
- Some women who have small, low-grade or intermediate-grade DCIS and widely negative surgical margins may choose to avoid radiation therapy altogether. (When DCIS is hormone receptor-positive, hormone therapy is taken.)
Radiation therapy for DCIS is usually given every day, 5 days a week, for 3-4 weeks. Some people may only need 5 radiation therapy treatments.
Learn about going through radiation therapy.
Learn more about surgical margins.
| For a summary of research studies on treatment for DCIS with a lumpectomy plus whole breast radiation therapy, visit the Breast Cancer Research Studies section. |
Hormone therapy
Hormone receptor status
A pathologist determines the hormone receptor status of the DCIS by testing the tissue removed during a biopsy.
- Hormone receptor-positive (estrogen receptor-positive/progesterone receptor-positive) DCIS tumors express hormone receptors. This means they have a lot of hormone receptors. Hormone receptor-positive tumors need estrogen and/or progesterone to grow.
- Hormone receptor-negative (estrogen receptor-negative/progesterone receptor-negative) DCIS tumors do not express hormone receptors. This means they have few or no hormone receptors.
Most cases of DCIS are hormone receptor-positive. People with hormone receptor-positive DCIS may benefit from hormone therapy (tamoxifen or an aromatase inhibitor) [5,9,13-17].
Learn about hormone receptor status and invasive breast cancer.
After a mastectomy
Because the risk of recurrence of DCIS is so low after a mastectomy, hormone therapy isn’t usually recommended for women who have a mastectomy for DCIS. They have an excellent chance of survival with a very low risk of DCIS recurrence or developing invasive breast cancer in the opposite breast [5].
For women who have a mastectomy for DCIS, the benefit of hormone therapy would likely be very small and would mostly affect the risk of cancer in the opposite breast.
After a lumpectomy
The National Comprehensive Cancer Network (NCCN) recommends women who are treated with a lumpectomy for hormone receptor-positive DCIS consider taking hormone therapy (tamoxifen or an aromatase inhibitor) for 5 years [5].
In women treated with a lumpectomy and radiation therapy for DCIS, studies have shown hormone therapy can lower the risk of [5,9,13-17]:
- DCIS recurrence (a return of DCIS)
- Invasive breast cancer
These risks are lowered in both the treated breast and the opposite breast.
Low-dose tamoxifen
Women who have a lumpectomy for hormone receptor-positive DCIS and have trouble taking tamoxifen due to the side effects may consider taking low-dose tamoxifen for 3-5 years [5].
Learn more about tamoxifen, including possible side effects.
Learn more about aromatase inhibitors, including possible side effects.
Learn more about factors that affect treatment options.
Learn about emerging areas in the treatment of DCIS.
| For a summary of research studies on tamoxifen for the treatment of DCIS, visit the Breast Cancer Research Studies section. |
Risk of developing invasive breast cancer after DCIS
After treatment for DCIS, there’s a small risk of:
- DCIS recurrence (a return of DCIS)
- Invasive breast cancer
These risks are higher with a lumpectomy plus radiation therapy than with a mastectomy [5]. However, overall survival is the same after either treatment [5].
Higher grade DCIS appears more likely than lower grade DCIS to recur as DCIS or invasive breast cancer after treatment (surgery, with or without radiation therapy) [18].
With close follow-up, invasive breast cancer is usually caught early and can be treated effectively.
Questions you may want to ask your health care provider
About your diagnosis
- Is DCIS breast cancer? How is DCIS different from invasive breast cancer?
- Is my DCIS hormone receptor-positive or hormone receptor-negative?
Treatment
- What are my treatment options?
- Which treatments do you recommend for me and why?
- What are my chances of having a recurrence of DCIS or developing invasive breast cancer?
- Should I have genetic testing before treatment begins?
- Do you recommend a lumpectomy for me? If not, why?
- Will I need radiation therapy after my surgery? If so, when will I meet with the radiation oncologist to discuss my radiation therapy?
- If I have a lumpectomy plus radiation therapy now, and the breast cancer returns (as a DCIS recurrence or invasive breast cancer), will I need to have a mastectomy at that time?
- If I have a mastectomy, will a sentinel lymph node biopsy be done?
- Were my tumor margins negative (also called uninvolved, clean or clear)? If not, what more will be done?
- Talk to me about breast reconstruction if I have a mastectomy. If I decide I want reconstruction, when can I have it (at the same time as the mastectomy or at a later date)? What are the risks? What about prosthesis options? Who else should I see to discuss and plan for reconstruction or prosthesis?
- If my DCIS is hormone receptor-positive, what are the benefits of taking a hormone therapy drug (tamoxifen or an aromatase inhibitor)?
- If I take hormone therapy, what do I need to consider before treatment begins if I would like to have a child after treatment?
- Are there clinical trials enrolling people with DCIS? If so, how can I learn more?
- How quickly do I need to make a decision about my treatment plan?
- Will some of the tissue removed during surgery be stored? Where will it be stored? For how long? How can it be accessed in the future?
Follow-up care
- How often will I have check-ups and follow-up tests after treatment ends?
- Will a follow-up care plan be prepared for me?
- Who’s in charge of my follow-up care?
Learn more about talking with your health care provider.
Learn about getting high-quality care, including choosing a doctor.
Susan G. Komen® resources
If you’ve been recently diagnosed with DCIS or feel too overwhelmed to know where to begin to gather information, it may be helpful to download and print some of Komen‘s resources. For example, we have Questions to Ask Your Doctor About Breast Cancer Surgery and Questions to Ask Your Doctor About Radiation Therapy and Side Effects.
We also have a Breast Cancer Diagnosis Discussion Guide which can help you gather information about your DCIS diagnosis, treatment options and more. You’ll also find a breast cancer glossary and a place to list members of your health care team.
You can download and print resources to take with you to your next doctor’s appointment, or you can save them on your computer, tablet or phone using an app such as Adobe. Plenty of space and a notes section are provided to write or type the answers to the questions.
There are other Questions to Ask Your Doctor resources on many different breast cancer topics you may wish to download.
Financial assistance
Costs related to DCIS treatment can quickly become a financial burden for you and your family. Dealing with finances and insurance can be overwhelming.
Many hospitals have financial counselors who can discuss insurance and cost coverage with you. They may be able to arrange a payment plan for hospital-related costs.
You may qualify for financial assistance from federal, state or local programs. A financial counselor or social worker at your hospital can help you learn about financial assistance programs.
Learn about insurance and financial assistance programs.
Learn about transportation, lodging, childcare and eldercare assistance programs.
Learn about resources that offer social support and practical support.
Komen Financial Assistance Program |
Susan G. Komen® created the Komen Financial Assistance Program to help those struggling with the costs of breast cancer treatment by providing financial assistance to eligible individuals. To learn more about this program and other helpful resources, call the Komen Patient Care Center at 1-877 GO KOMEN (1-877-465-6636) or email helpline@komen.org. Se habla español. Learn about other financial assistance programs. |
Clinical trials
Research is ongoing to improve all areas of treatment for DCIS.
New therapies are being studied in clinical trials. The results of these studies will determine whether these therapies become part of the standard of care. Clinical trials can also study other parts of care, such as ways to manage side effects.
After discussing the benefits and risks with your oncologist, we encourage you to consider joining a clinical trial if there’s one right for you.
Read our blog, Four Things to Know About Clinical Trials When You’re Diagnosed with Breast Cancer.
Susan G. Komen® Patient Care Center |
If you or a loved one needs information or resources about clinical trials, the Patient Care Center can help. Contact the Komen Breast Care Helpline at 1-877-465-6636 or email clinicaltrialinfo@komen.org. Se habla español. |

BreastCancerTrials.org in collaboration with Komen offers a custom matching service to help find clinical trials that fit your health needs.
Learn what else Komen is doing to help people find and participate in breast cancer clinical trials, including trials supported by Komen.
Learn how Komen-funded research is improving treatment for breast cancer.
Learn more about clinical trials.
You’re not alone
If you’ve been diagnosed with DCIS, it’s normal to feel many different emotions. You may be worried or scared about going through treatment and the side effects you may have. You may also be thinking about what your life will be like after you finish treatment.
Many people have been where you are today. They had the same concerns and fears. They’ve gone through treatment for DCIS and are adjusting to life after it.
It may be helpful for you to talk about how you’re feeling and get support from others. Having people in your life who can relate to some of what you’re going through may help you feel less alone.
You could share your experience and advice with others going through treatment for DCIS or those who’ve finished treatment. You can do this in a support group or by connecting one-on-one with another DCIS survivor. A social worker or patient navigator can help you find these resources.
You can also talk with your health care providers about how you’re feeling. They care about your overall well-being and want to help They may connect you to a mental health provider on your health care team, such as a social worker, for emotional support.
Our Support section has a list of resources to help find local and online support groups and other resources.
Learn more about social support and support groups.
Learn about healthy ways to cope with stress.
Susan G. Komen® Support Resources |
|
Updated 03/23/26
This content is regularly reviewed by an expert panel including researchers, practicing clinicians and patient advocates.




