What’s New in Breast Cancer
This section gives an overview of new breast cancer treatment breakthroughs and recent developments in research that are fueling new ways to assess risk, and prevent, detect, diagnose and treat breast cancer. Advances in breast cancer care are evaluated through a rigorous process that includes clinical trials and regulatory approvals before being considered standards of care and included in breast cancer care guidelines. Komen’s research team monitors the rapidly evolving breast cancer landscape, and here we will highlight new breast cancer treatment breakthroughs, innovations in technology or key advances that may be added or are new to guidelines. We will share these research advancements to empower patients with knowledge to help them make informed decisions with their doctors.
Use these links to jump to the topics below.
Treatments and Drugs
For patients, new treatments can mean more options and more hope. Researchers are working to develop new breast cancer treatment breakthroughs, such as more effective drugs that will specifically target breast cancer cells, minimize side effects and prevent breast cancer cells from coming back. While some treatments increase the effectiveness of existing drugs, others may offer new, innovative strategies for attacking tumor cells.
As of April 2026, the following new treatments and drugs are currently in clinical trials and have not yet received approval from the Food and Drug Administration (FDA), meaning they have not yet been reviewed for safety and effectiveness for their intended use.
Giredestrant: An investigational oral SERD for early‑stage hormone receptor‑positive breast cancer
Giredestrant is an oral selective estrogen receptor degrader (SERD), a type of hormone therapy that works by breaking down the estrogen receptor in hormone receptor-positive (HR+) breast cancer cells. In the phase 3 lidERA trial, giredestrant improved disease-free survival compared with standard hormone therapy. If approved, it could become a new oral hormone therapy option for some people with early-stage HR+/HER2-negative (HER2-) breast cancer and mark the first oral SERD to benefit people with early breast cancer.
Zanidatamab and chemotherapy: A new HER2-targeted approach following T-DXd
The EmpowHER-303 trial is an ongoing study evaluating zanidatamab with chemotherapy for HER2-positive (HER2+) metastatic breast cancer (MBC) after disease progression on trastuzumab deruxtecan (T-DXd). Zanidatamab, a bispecific antibody, is different from existing HER2-targeted therapies because it can bind to two different parts of the HER2 protein at the same time instead of just one. This may block cancer growth more completely than medications that attach to just one part, which may help meet a growing need for new treatment options for people with HER2+ MBC whose disease has progressed after T-DXd, as more individuals receive this therapy earlier in treatment (see below in Approaches to Care).
Learn more in this clip from our Breast Cancer Breakthroughs series.
RLY-2608: Targeting breast cancer mutations
About 40% of HR+ metastatic breast cancers have mutations in the PIK3CA gene. The mutated protein that arises from PIK3CA mutations promotes growth of the cancer cells. RLY-2608 is a new drug that specifically blocks the mutant protein from driving cancer growth. By blocking only the mutant protein and not the normal protein, it’s possible that this drug may have fewer unwanted side effects. Early results showed that RLY-2608 combined with fulvestrant (a hormone therapy drug) led to a median of 10.3 months before participants’ metastatic breast cancer progressed. A phase 3 trial testing RLY-2608 in combination with fulvestrant is now underway.
Learn more in our Breast Cancer Breakthroughs installment “On the Horizon of PI3K Pathway Inhibition.”
Technology
New and improved technologies may be able to increase the speed and accuracy of detecting, diagnosing or monitoring breast cancer for progression and response to treatment.
ctDNA & MRD testing: Expanding applications and new trials
The use of circulating tumor DNA (ctDNA) and minimal residual disease (MRD) testing is expanding across cancer research and care, including in breast cancer. New clinical trials continue to explore applications for ctDNA in breast cancer care such as detecting recurrence, helping inform potential treatment decisions in specific scenarios and monitoring treatment responses – though limitations in sensitivity, standardization and clinical value remain. Importantly, routine ctDNA-guided care is still not in the National Comprehensive Cancer Network’s (NCCN) Clinical Practice Guidelines in oncology for most patients. The CATE clinical trial is one new trial aiming to use ctDNA testing to proactively identify early-stage HR+/HER2- breast cancers at high risk of recurrence, and adjust treatment based on that risk. Read more about the CATE clinical trial here.
Artificial Intelligence (AI)-based risk assessment tool
Predicting one’s risk for developing breast cancer currently relies on several different factors including a person’s gender, age, ethnicity, reproductive history, health background and family health history. A new AI-based risk-assessment technology was recently granted authorization from the FDA. While the FDA has previously approved AI tools to assist in breast cancer detection and risk management, this is the first to receive authorization specifically for predicting five-year risk directly from a screening mammogram. However, the use of AI to provide additional information about someone’s breast cancer risk from mammograms is still being studied and is not yet part of standard screening care for most people.
Tailoring breast cancer screening to a person’s unique risk of breast cancer
The WISDOM trial is helping to answer whether tailoring breast cancer screening to a person’s individual risk is as safe and effective as recommending annual mammograms starting at age 40. In its first phase, WISDOM 1.0, women ages 40 to 74 were enrolled. Early results show that a more personalized approach to screening, incorporating factors like family history, breast density, genetic information and more can detect cancers that are stage 2B or higher as safely as annual mammography.
Learn more about the WISDOM study in our Highlights from SABCS 2025 installment of Breast Cancer Breakthroughs.
Oncotype DX Breast Recurrence Score® test updates
The Oncotype DX Breast Recurrence Score test is a tumor profiling test used for some people with early-stage HR+, HER2-, lymph node-negative breast cancer to help understand their risk of cancer returning and whether there’s a benefit to giving chemotherapy in addition to hormone therapy. Based on updated data from the TAILORx trial, the test now provides more tailored information based on age for people whose cancer is lymph node-negative.
Approaches to Care
With knowledge gained from clinical trials, researchers are seeking new ways to improve patient outcomes while using existing drugs. Some new breast cancer treatment breakthroughs are the result of combining certain drugs, finding which patients can skip certain elements of treatment or changing the order of their treatments to maximize effectiveness or to minimize side effects.
Trastuzumab deruxtecan (T-DXd) shifting earlier in HER2+ breast cancer treatment
Several DESTINY-Breast clinical trials have tested if T-DXd (an antibody-drug conjugate or ADC) is effective when used earlier in the treatment of HER2+ breast cancer.
- DESTINY-Breast05 demonstrated that T-DXd improved outcomes for people with high-risk residual HER2+ early breast cancer following neoadjuvant therapy compared to ado-trastuzumab emtansine (T-DM1) (an ADC).
- Destiny-Breast11 showed that when T-DXd was used as part of neoadjuvant therapy, it led to a higher pathologic complete response rate compared to chemotherapy-based approaches in HER2+ early breast cancer.
- DESTINY-Breast09 also evaluated T-DXd as a first-line option for HER2+ MBC and is now FDA-approved in combination with pertuzumab in this setting.
Together these trial results could shift how (and, more so, when) ADC’s might be used not only in metastatic disease but also earlier in the course of HER2+ breast cancer treatment.
Adding tucatinib to first-line maintenance therapy in HER2+ metastatic breast cancer (MBC)
In the HER2CLIMB-05 trial, adding tucatinib to trastuzumab and pertuzumab (all HER2-targeted therapies) as first-line maintenance therapy after induction (or initial treatment period on chemotherapy) improved progression-free survival. This supports a new maintenance strategy designed to extend the time people can avoid chemotherapy following initial treatment.
TROP2-targeting antibody drug conjugates (ADCs) become a first‑line option for some metastatic triple-negative breast cancers(TNBC)
The results of recent trials are impacting how doctors treat metastatic TNBC – particularly for people who cannot receive immunotherapy. ADCs like sacituzumab govitecan (SG) and datopotamab deruxtecan (Dato-DXd) have shown they can help people live longer without disease progression compared to standard chemotherapy in first-line metastatic TNBC based on the TROPION-Breast02, ASCENT-03 and ASCENT-04 clinical trials.
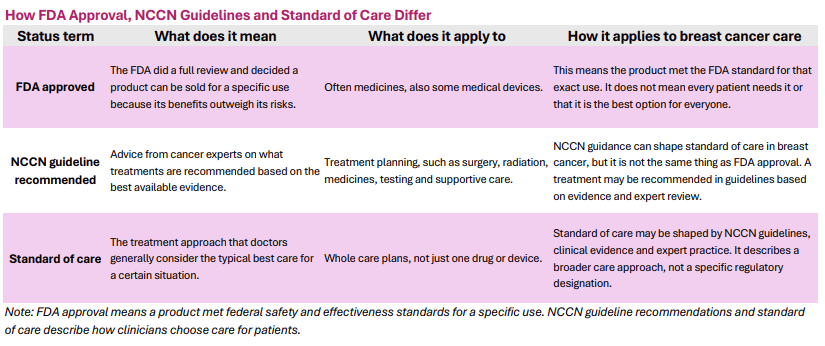
The NCCN guidelines now list SG and Dato-DXd among the recommended first-line treatment options for certain populations with metastatic TNBC even though these drugs have not yet received FDA approval in this specific setting. This points to how quickly ADCs are moving into earlier lines of treatment, offering at times more effective options than traditional chemotherapy for some people with metastatic TNBC.
Learn more in our recent installment of Breast Cancer Breakthroughs: What’s New for Metastatic TNBC?
Komen will be closely monitoring the results of these studies and more at upcoming scientific conferences. We hope to see more promising data regarding new ways to prevent, detect, diagnose and treat breast cancer.
Last Updated April 2026






